Quantum Mechanics 1Mark Questions
- State and briefly explain two applications of quantum mechanics in modern technology.
• Semiconductors: basis of modern electronics like computers, mobiles, and solar cells.
• Lasers: based on stimulated emission, applied in medicine, communication, and industry. - Using the de-Broglie hypothesis, determine the wavelength of a particle given its momentum.
The wavelength is given by \( \lambda = \dfrac{h}{p} \), where \( h \) is Planck’s constant and \( p \) is the momentum.
- Explain Heisenberg's Uncertainty Principle.
Heisenberg's Uncertainty Principle states that it is impossible to simultaneously know the exact position (\( x \)) and momentum (\( p \)) of a particle with perfect precision, mathematically \( \Delta x \cdot \Delta p \geq \frac{\hbar}{2} \), where \( \hbar \) is the reduced Planck's constant. This arises from the wave-particle duality.
- What is wavefunction? Write two of its properties.
The wavefunction \( \psi \) describes a particle's quantum state.
• \( |\psi|^2 \) gives probability density.
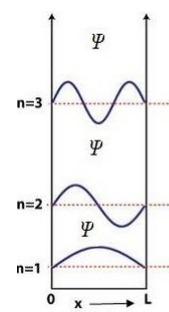
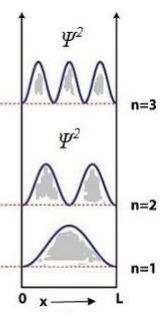
• \( \psi \) must be finite, single-valued, and continuous. - Draw the wavefunction and probabilityof the particle for energy levels (n = 1, 2, 3) in one-dimensional potential box.
For a box of length \( L: \)
\( \psi_n(x)=\sqrt{\tfrac{2}{L}}\sin\left(\tfrac{n\pi x}{L}\right) \) and \( P_n(x)=|\psi_n|^2 \).
• n=1: one half-wave, no node inside.
• n=2: two half-waves, one node inside.
• n=3: three half-waves, two nodes inside.
- Differentiate between eigenvalue and eigenfunction.
• Eigenvalue: measurable number (e.g., energy).
• Eigenfunction: wavefunction corresponding to that eigenvalue. - Why does a particle in a box never have zero energy?
Zero energy means exact position and zero momentum, violating the uncertainty principle. Hence ground state energy is always > 0.
- State the type of materials to which Bloch’s theorem applies.
Bloch’s theorem applies to periodic crystalline materials.
- Why are Bloch functions important for electron motion in solids?
Bloch functions describe electron states in periodic potentials and explain band formation and conductivity.
- Define Brillouin zone.
It is the primitive cell in reciprocal (momentum) space of a crystal lattice.
- Why do band gaps arise in solids according to Kronig-Penney model?
Band gaps appear due to electron wave interference in periodic crystal potentials, making some energies forbidden.
- Why is effective mass important in solid-state physics?
It describes how electrons respond to forces in a crystal and simplifies transport calculations in devices.
- Compare band gaps in conductors, semiconductors, and insulators.
• Conductor: no gap, bands overlap.
• Semiconductor: small gap (~1 eV).
• Insulator: large gap (>3 eV). - How are solids classified based on band theory?
Solids are classified as conductors, semiconductors, or insulators depending on their band gap size.
- Differentiate between continuous and discrete energy spectrum.
• Discrete: specific energy levels (atoms, particle in a box).
• Continuous: unbroken range of energies (free particles).