Sol-Gel Method – Nanomaterial Synthesis
Sol-Gel Method is a bottom-up chemical technique for synthesizing nanomaterials in which a liquid colloidal solution (sol) is transformed into a solid network (gel) through controlled hydrolysis and condensation reactions, allowing precise control over particle size and composition.
The sol-gel method is a widely used bottom-up nanomaterial synthesis technique that begins with precursor chemicals dissolved in a solvent to form a colloidal sol. Over time, the sol undergoes hydrolysis and condensation reactions to produce a gel network. After drying and heat treatment, the resulting solid material can be used for a variety of applications including coatings, ceramics, thin films, and nanoparticles.
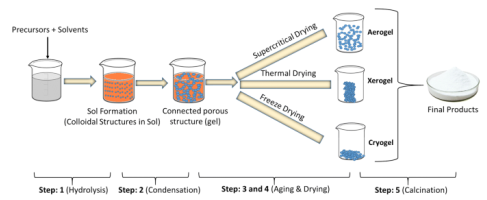
Working Principle and Steps of the Sol-Gel Process
The sol-gel process involves several controlled stages that convert liquid precursors into a solid nanostructured material through chemical reactions.
Sol Preparation
Suitable precursors are dissolved in water or alcohol to form a homogeneous colloidal solution called a “sol.” Hydrolysis of metal alkoxide precursors is common in this stage.
Gelation
The sol particles begin to link through condensation reactions, gradually forming a three-dimensional solid network known as a gel.
Drying and Aging
The wet gel retains solvent within its pores; controlled drying removes the solvent and densifies the network while minimizing cracking.
Heat Treatment / Calcination
The dried gel is heated to remove residual organics and enhance structural stability. This step also defines the final phase (e.g., oxide) and porosity of the material.
Advantages of Sol-Gel Method
- Produces high-purity products with controlled composition and nanostructure.
- Effective control over particle size and morphology by tuning reaction conditions.
- Operates at relatively low temperatures compared to many other synthesis routes.
- Enables uniform coating of complex surfaces and thin films.
- Versatile for a wide range of materials including oxides and composites.
Applications of Sol-Gel Method
- Thin films and coatings in optics and electronics.
- Ceramic and glass nanomaterials for structural applications.
- Nanoparticle production for sensors and catalysts.
- Porous materials such as aerogels and xerogels.
Conclusion
The sol-gel method is a versatile and widely used bottom-up chemical synthesis route for nanomaterials, providing excellent control over composition, size, and structure. It is especially useful in applications requiring uniform coatings, high purity, and tailored nanostructures.
Related topics: Synthesis of Nanomaterials | Introduction to Nanotechnology | Applications of Nanotechnology