Einstein Coefficients
Lets consider two levels of an atomic system as shown in Fig. 1 and let N1 and N2 be the number of atoms per unit volume present in the energy levels E1 and E2, respectively. An atom in the lower energy level E1 can absorb the incident radiation at a frequency v and be excited to E2.
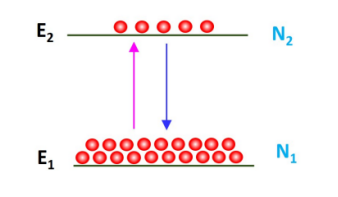
Figure.1. Energy diagram of stimulated absorption, spontaneous emission and stimulated emission
The rate (R1) at which absorption takes place from E1 to E2 will be proportional to the number of atoms present in the level E1 and also to the energy density of the radiation and let it be u(v).
\[R_1 \propto N_1 u(v) \rightarrow R_1 = B_{12} N_1 u(v) \tag{1} \]Where, B12 is a constant of proportionality.
Now during the de-excitation process from E2 to E1, Einstein postulated that an atom can make a transition through two distinct processes, namely stimulated emission and spontaneous emission.
In the case of stimulated emission, the radiation which is incident on the atom stimulates it to emit radiation and the rate of transition to the lower energy level is proportional to the energy density of radiation at the frequency v.
Thus, the number of stimulated emissions per unit time per unit volume will be
\[ R_2 = B_{21}N_2u(v) \tag{2}\]For spontaneous emission of atoms from higher energy levels, the rate will depend on the number of atom in level E2, Hence
\[ R_3 = A_{21}N_2 \tag{3}\]At thermal equilibrium, the number of upward transitions must be equal to the number of downward transitions,
\[ R_1 = R_2+R_3 \]Substitute R1, R2 and R3 values in the above equation,
\[ B_{12} N_1 u(v) = B_{21}N_2u(v) + A_{21}N_2 \ \tag{4} \]On solving,
\[ u(v) = \frac{A_{21}}{ \frac{N_1}{N_2} B_12 - B_21 } \tag{5}\]Using Boltzman’s Law, the equilibrium population levels at E1 and E2 is given by
\[ N_1 = \exp\left(-\frac{E_1}{kT}\right) \quad \text{and} \quad N_2 = \exp\left(-\frac{E_2}{kT}\right) \] \[ \frac{N_1}{N_2} = \exp\left(\frac{E_2 - E_1}{kT}\right) = \exp\left(\frac{h\nu}{kT}\right) \tag{6} \]substitute the above Eqn in Eqn 6,
\[ u(\nu) = \frac{A_{21}/B_{21}}{B_{21} \exp\left(\frac{h\nu}{kT}\right) - 1} \tag{7} \]According to Planck’s law, the radiation energy density per unit frequency is
\[ u(\nu) = \frac{8\pi h \nu^3}{c^3 \left(\exp\left(\frac{h\nu}{kT}\right) - 1\right)} \tag{8} \]Comparing Eqn 7 and Eqn 8,
\[ B_{12} = B_{21} = B \] and \[ \frac{A_{21}}{B_{21}} = \frac{8\pi h \nu^3}{c^3} \tag{9}\]Thus the stimulated emission rate per atom is the same as the absorption rate per atom. The coefficients A and B are referred to as the Einstein A and B coefficients.
At thermal equilibrium, the ratio of number of spontaneous to stimulated emissions is given by
\[ R = \frac{R_3}{R_2} = \frac{A_{21} N_2}{B_{21} N_2 u(\nu)} \]Substituting the value of A21/B21 from 9 and u(v) from Eqn 7,
\[ R = \exp\left( \frac{h\nu}{kT} - 1 \right) \]